HE PECULIARITY OF THE RIGHT-HEMISPHERE FUNCTION IN DEPRESSION:
SOLVING THE PARADOXES
Vadim S. Rotenberg, E-mail: vadir@post.tau.ac.il,
Abarbanel Mental Health Center, Sackler Medical School, Tel-Aviv University, Keren Kayemet Str. 15, Bat-Yam, Tel-Aviv, Israel
Progress in Neuro-Psychopharmacology and Biological Psychiatry
Volume 28, Issue 1
January 2004,
Pages 1-13
Accepted 11 July 2003. ; Available online 3 September 2003.
AbstractDepression is characterized by functional insufficiency of the right hemisphere combined with its physiological overactivation. This paradox can be solved in the frame of the general concept of brain laterality. According to the present assumption, the left hemisphere organizes any information in an unambiguous monosemantic context, and this process requires an additional activation of the brain cortex in order to restrict natural relationships between objects and events. On the contrary, the right hemisphere organizes any information in the polysemantic context based on the simultaneous capture of the numerous natural relationships between elements of information. In healthy creative subjects this process does not require additional physiological activation of the cortex. In depression the physiological overactivation of the right hemisphere reflects the unsuccessful effort to overcome its functional insufficiency.
Author Keywords: Cortical activation; Depression; Hemisphere functions; Mono- and polysemantic contexts
Abbreviations: ECT, electroconvulsive therapy; NA, negative affect

1. IntroductionThe present paper discusses a very complicated and contradictory topic: the relationship between functional activity and physiological activation of the right hemisphere in depression. In the first part of this paper the author presents a review of the literature related to this topic. In the second part of the paper a new concept of brain laterality is proposed. In the final part of the paper this new concept is applied to the right-hemisphere function in depression. 2. Functional brain asymmetry in depression: data and conceptsAccording to contemporary thinking, functional brain asymmetry substantially contributes to the psychophysiological mechanisms of affective disorders [Davidson, 1992, Davidson, 1993, Flor-Henry, 1983 and Heller, 1993]. [Schwartz et al., 1975] have shown that greater right- than left-hemisphere activation mediates the effect of negative affect. It is worth emphasizing that the relative hyperactivation of the frontal pole of the right hemisphere characterizes not only major depression [Debener et al., 2000, Bench et al., 1992 and Bruder et al., 1989] but also dysthymia [Henriques and Davidson, 1990] as well as depressed mood induced in normal subjects [Tucker et al., 1981, Tomarken et al., 1990 and Hagemann et al., 1999]. Thus, it is a characteristic of the state independent of nosology. Many investigations have shown that organic damage of the left hemisphere is usually accompanied by depressed mood and "catastrophic reaction," whereas right-hemisphere damage is associated with euphoric or "indifference" reaction [Finset, 1988, Gainotti, 1972 and Starkstein and Robinson, 1988]. According to [Starkstein and Robinson, 1988], the closer the brain lesion to the left frontal pole, the higher the incidence and severity of depression. These data correspond to the emotional outcome of the intracarotid amobarbital test or sodium amytal test applied to the left or right hemisphere of depressed patients [Lee et al., 1988 and Perria et al., 1961], as well as to the outcome of unilateral electroconvulsive therapy (ECT) used in the treatment of depression [Galin, 1974]. All the abovementioned data lead to the hypothesis that the left hemisphere is specialized for the maintenance of pleasurable affect, whereas the right hemisphere is responsible for the formation of the unpleasurable affect [Heller, 1990 and Heller, 1993]. According to this hypothesis, when the function of one hemisphere is suppressed (by organic damage, ECT, or sodium amytal) the emotional state and observed behavior reflect the functioning of the intact hemisphere that is not balanced by the functioning of the suppressed one. Another approach to the same data was proposed by [Tucker, 1981], who assumed that the observed emotional state reflects the disinhibition of the ipsilateral subcortical area of the damaged hemisphere. According to [Tucker, 1981], negative affect reflects the disinhibited activity of the left subcortical area and positive affect reflects the disinhibited activity of the right subcortical mechanisms. However, the damage of the hemisphere caused by stroke is often broad enough to include not only the cortical but also the subcortical area. It was shown that left subcortical lesions are also accompanied by depression ( [Liotti et al., 1988], cited in [Tucker, 1988]). Moreover, severity of depression was related to the depth of lesion [Finset, 1988], and left anterior subcortical lesions are significantly associated with major depression [Starkstein and Robinson, 1988]. The same is correct for the sodium amytal injected in carotid vessels: the latter supply with blood not only the superficial cortical area, but also the deep subcortical area. It is also difficult to imagine ECT not involving the subcortical area. Thus, the initial approach, which explains an emotional state by the suppression of one hemisphere and by the nonbalanced increased activity of the opposite hemisphere, seems to have more supporting arguments. 3. The right frontal physiological activity in depression: experimental dataMost investigations show a relative increase of the electrical activity of the right frontal lobe and prefrontal cortex in depression [Heller, 1990, Henriques and Davidson, 1990, Jacobs and Snyder, 1996 and Schaffer et al., 1983]. It was shown [Davidson et al., 1990 and Tucker et al., 1981] that depressed mood and the experience of disgust are accompanied in normal subjects by the right-sided frontal activation compared with euphoric mood and the experience of happiness. Subjects with higher right-sided frontal activation reported more negative affects than subjects with higher left-sided frontal activation [Hagemann et al., 1999]. The left frontal cortical activation was shown to be higher during reward trials than during punishment [Sobotka et al., 1992]. The resting alpha power in the frontal area being less on the right side predicted self-reported negative affect in response to negative film clips [Tomarken et al., 1990 and Tomarken et al., 1992]. Subjects with reports of positive affect in response to positive film display greater left frontal activation, while subjects with reports of negative affect in response to negative film display greater right frontal activation [Wheeler et al., 1993]. A relative left frontal hypoactivation in depression was confirmed in most investigations with only few exceptions [Debener et al., 2000]. It is a discussion on the role of ventromedial and dorsolateral parts of the prefrontal cortex in emotional regulation and particularly in depression. According to [Davidson and Irwin, 1999], the ventromedial part is involved in the representation of elementary positive and negative affective states in the absence of immediately present incentives, whereas the dorsolateral part is involved in the representation of goal states toward which these more elementary positive and negative states are directed. In some investigations on major depression a reduced activity of the dorsolateral prefrontal cortex was found to correlate with the severity of depression, psychomotor retardation or cognitive disturbance [Soares and Mann, 1997, Drevets, 1998, Bench et al., 1993 and Dolan et al., 1993], whereas the ventral prefrontal cortex showed increased activity [Drevets et al., 1992]. In other investigations, brain metabolism was either increased in all parts of the left and right prefrontal cortex [Brody et al., 2001] or there was no definite alteration of metabolism in these parts of the cortex [Martin et al., 2001]. Thus, data in this topic are very contradictive and it is now difficult to discuss the function of different parts of the prefrontal cortex in the context of brain laterality. It is important to emphasize that the negative affect (depression) is related almost exclusively to the activation of the frontal region of the right hemisphere. In the central, temporal, and posterior regions, depressed patients display a relatively higher activation on the left side of the brain than on the right side [Hagemann et al., 1998, Perris et al., 1981 and Tucker and Dawson, 1984]. Healthy subjects subjected to relative left-sided anterior temporal activation reported more intensive negative affect in response to negative slides than subjects with right-sided anterior temporal activation [Wheeler et al., 1993]. Unhappy males scoring high on the negative affect (NA) as well as unemotional females demonstrated activation of the left anterior temporal region, whereas subjects scoring low on NA (happy females and unemotional males) demonstrated activation of the right anterior temporal region [Hagemann et al., 1999]. Alpha activity in posterior regions was blocked when a good target appeared in the left visual field and a bad target in the right visual field [Tucker et al., 1999]. 4. Affect and brain laterality: speculations and contradictionsThe theoretical integration of all the abovementioned experimental data is far from being comprehensive and exhaustive. A very substantial analysis performed by [Heller and Nitschke, 1998] led them to the conclusion that the anterior regions of the brain consistently emerge as important in affective valence related to a person's affective style, whereas the right parietotemporal region is important in arousal. [Davidson, 1998] presented a broader and very well known interpretation of the relationships between negative affect and right frontal and positive affect and left frontal physiological activation. According to [Davidson, 1998], the left frontal area is responsible for the approach behavior whereas the right frontal area is responsible for the withdrawal behavior. Approach is related to positive emotions such as enthusiasm and pride, whereas withdrawal is related to negative affect such as fear and disgust. However, the conclusion that in healthy adult subjects the higher activity of the right hemisphere is related to the negative affect/withdrawal behavior, and the higher activity of the left hemisphere is related to the positive affect/approach behavior, seems to be in contradiction with other data from behavior rarely discussed in the context of these concepts––creativity and empathy. First, the right hemisphere plays an important role in solving insight problems [Bowden and Beeman, 1998], i.e., creativity, and creativity is without doubt an approach behavior. According to the definition [Kuper, 1988], creativity is the ability to bring something new into existence, and the creative new product has its roots in reality, in the covert connections and interrelationships between objects, events, and ideas. Ingredients of the creative way of thinking include the following: seeing things in a new way, making connections, taking risks, being alert to chance and to the opportunity presented by contradictions and complexities, and recognizing familiar patterns in the unfamiliar. Thus, it is obvious that creative activity includes an approach to reality. Second, recognizing emotions from visually presented facial expression is also within the competence of the right hemisphere and requires somatosensory related cortices [Adolphs et al., 2000]. Subjects recognize another individual's emotional state by internally generating somatosensory representations that simulate how the other individual would feel when displaying a certain facial expression [Adolphs et al., 2000]. This corresponds to the notion of empathy, and empathy in any case is not related to avoidance behavior. Furthermore, creative mental activity is usually accompanied by pleasant affect. Pleasant affect facilitates creative problem solving and flexibility [Heller and Nitschke, 1998]. It was shown that the increase of the right-hemisphere contribution to the person's cognitive functioning correlates with a more positive affect [Tucker, 1988]. Subjects with a more right than left cognitive style reported less anxiety, less depression and showed more positive self-report. In the process of task performing, subjects with higher right-hemisphere activation rated themselves as having done better than they have done in reality [Tucker, 1988]. Those subjects who described themselves as more intuitive and nonverbal (domination of the right hemisphere) were also more likely to describe themselves in positive terms. Subjects who considered themselves to be analytic and verbal (left-hemisphere style of thinking) described themselves in negative terms [Swenson and Tucker, 1983]. Positive affect was associated with an increased tendency to use intuitive rather than analytical and logical strategies for problem solving [Isen et al., 1982]. Subjects with enhanced performance for the right visual field (left hemisphere) in identification of syllables displayed more pessimistic biases in rating performance, in comparison to subjects with enhanced performance for the left visual field (right hemisphere) [Levy, 1983]. As a result, it is difficult to conclude that in healthy subjects the activity of the right hemisphere is related to the negative affect and to withdrawal behavior. In addition, bipolar patients in manic state that according to the mood is opposite to depression display a left ear (right hemisphere) advantage in verbal dichotic listening for pair of digits demonstrating a functional overactivation of the right hemisphere [Karpinis et al., 1995]. 5. A main contradiction: disturbed right-hemisphere functions in depressionAlthough the right frontal lobe is physiologically hyperactivated in depression, the right-hemisphere functions are disturbed in depressed patients. According to blood flow studies, depression is characterized by the reduced right anterior brain functions [Baxter et al., 1989 and Bench et al., 1992]. The experience of sadness is characterized by the decreased blood flow in the right dorsolateral prefrontal and inferior parietal regions while recovery from depression is accompanied by the restoration of blood flow in the same regions [Mandal et al., 1999]. Depressed people are poor problem solvers, and unpleasant affect is associated with a decrease in the cognitive processes associated with left and right frontal lobe functions [Hartlage et al., 1993 and Heller and Nitschke, 1997]. According to [Banich, 1997], [Banich et al., 1992], and [Heller and Nitschke, 1997 and Heller and Nitschke, 1998], cognition (mental activity) in depression is characterized by deficits in the following right-hemispheric functions: ability to interpret the nonverbal information (e.g., facial expression, voice prosody, gesture), face recognition, nonverbal Wechsler performance, judgement of line orientation, design fluency, three-dimensional constructional praxis, spatial association learning, and performance with the nondominant hand on the Tactual Performance Test. Depressed patients exhibit a selective decrease of blood flow to right temporal and parietal regions when engaged in repetitive learning tasks. They demonstrate also impaired performance in tests on attention and concentration, while the neural substrate of the sustained attention to the external environment is localized in the dorsolateral prefrontal cortex [Liotti and Mayberg, 2001]. After induction into a depressed mood, but not euphoric mood, normal individuals exhibit a deficit in visual imagery and right ear attentional bias on a tone judgement task [Banich, 1997]. The visuospatial deficit in depression was confirmed by [Miller et al., 1995]. In a depressed mood, in comparison to the emotionally neutral state, subjects display a slowing of reaction time to material presented in the left visual field. Presentation of the material in the right visual field was not accompanied by slowing of the reaction time [Liotti et al., 1988]. Depression was characterized by less vivid imagery [Tucker et al., 1981], while alleviation of the depressed mood led to the improvement of right-hemisphere task performance [Kronfol et al., 1978]. Alexithymia and poverty of dream recall are also related to decreased right-hemisphere functioning [Rotenberg, 1995]. Depressed patients, like right-brain-damaged patients, failed to recall the central aspects of the story, but recalled more specific items and were unable to put information in a larger social and environmental context [Gardner et al., 1983]. Depressed patients demonstrate poor right-hemisphere performance for all types of stimuli [Bruder et al., 1989]. In subjects with a sad affect, reaction time was slowed for the right hemisphere [Ladavas et al., 1984]. All the preceding data confirm the decreased functional ability of the right hemisphere in contradiction to the physiological hyperactivation of its frontal region. One possible approach for solving this contradiction is proposed by [Tucker et al., 1981]. According to this proposition, the negative affective state, which corresponds to the physiological hyperactivation of the anterior region of the right hemisphere, competes for processing capacity with the cognitive operations in the right hemisphere. Such competition seems to be quite possible. However, at least one question remains open with this explanation: Why the functional disability of the right frontal region caused by the competition between cognitive functions and depressive affect (which actually affects the whole brain) is accompanied by the physiological hyperactivity of only the frontal part of the right hemisphere, while the functional deficit of the left frontal region, although also present (for instance, in children of depressed mothers; [Dawson, 1994 and Field et al., 1995]) is not accompanied by such physiological hyperactivation. Another approach to this contradiction is based on the functional difference between the anterior and posterior right-hemisphere areas. According to this approach, physiological activation in depression is displayed only in the anterior lobe of the right hemisphere, which is not related to the abovementioned disturbed functions, while the parietotemporal part of the right hemisphere related to these functions is characterized by decreased physiological activity [Heller and Nitschke, 1997]. Metabolic activity in the right prefrontal cortex and in the right temporal areas in depression is also lower than in healthy subjects and in depressed patients after the successful treatment [Mayberg et al., 1999 and Wu et al., 1999]. This explanation may be partly relevant; however, it also conflicts with some experimental data. Whereas many deficient right-hemisphere functions in depression are performed by temporal, central, and occipital parts of the brain that are less activated than the frontal region and the symmetric regions of the left brain in depression, they are less activated as the same regions in the right brain of healthy subjects in states of happiness. However, some very important functions being deficient are related directly to the hyperactive frontal regions. According to [Banich, 1997], in healthy subjects the right anterior region is involved in design fluency and recency judgements. This area is very important for flexible and creative problem solving. The right anterior region is responsible for the development of problem-solving strategies in unfocused "open" conditions, which do not require a definite and single strategy. In such conditions, depressed people perform more poorly than nondepressed people and use a smaller amount of available relevant information. Depressed persons need to be provided by the definite problem-solving strategy they are following in a very rigid way [Weingartner et al., 1981]. Depressed patients are not flexible in their mental skills and less creative than normal people, and all these functions are related to the frontal lobe of the right hemisphere. Depressed mood reduces the retrieval of episodic memory [Ellis et al., 1985], which is related to the right anterior region [Heller and Nitschke, 1997]. Thus, although the right anterior region is physiologically activated in depression, it is functionally hypoactive and insufficient. Physiological activation does not conform to the functional activity. This problem has to be discussed in more detail. 6. Electroencephalogram (EEG) and brain functional efficacyAccording to [Lindsley, 1951], the relationship between physiological activity and functional efficacy of the brain, and especially of the right hemisphere, is in general (in healthy subjects) not so linear as it is expected to be. In some particular conditions the right hemisphere displays an obvious discrepancy between bioelectrical activation and functional efficacy. It was shown that the process of imagination in subjects with the domination of the right hemisphere occurs without additional desynchronization (decrease of alpha index) on the EEG [De Pascalis and Palumbo, 1986, Grossman, 1988 and Jones-Gotman and Milner, 1977]. The alpha index reflects the level of the nonspecific activation caused by the brain stem reticular formation, and it reflects a negative correlation between the alpha index and the degree of involvement of the corresponding cortical area in mental activity [Wertheim, 1974]. Altered states of consciousness (samadha-yoga, Zen-meditation) characterized by the relative domination of the right hemisphere [Ornstein, 1972 and Rotenberg and Arshavsky, 1991] are characterized also by increased alpha activity [Hirai, 1974, Kasumoto and Hirai, 1966 and Stigsby et al., 1981]. [Martindale, 1975] and [Whitton, 1978] have shown that creative subjects demonstrate an increase in alpha activity, especially in the right hemisphere, in the process of the solution of nonroutine tasks. At the same time, these subjects demonstrated decrease of alpha index in the process of the solution of routine logical tasks. Noncreative subjects displayed EEG desynchronization in the process of the solution of routine as well as creative tasks. Gifted adolescents display an advanced level of alpha activity at different (including frontal) locations of the right hemisphere, in comparison to peers with average mental skills [Alexander et al., 1996], and the authors emphasized that this right-hemisphere inactivity in gifted subjects is in contrast to the previous studies that indicated greater increase of activity at the same locations during formal cognitive tasks. In representatives of the Eastern culture, with the right-hemisphere cognitive set, the increase of the spatial synchronization (which reflects the increase of functional activity [Livanov and Sviderskaya, 1984] occurs in the right hemisphere in the process of imagination, without additional decrease of alpha index [Rotenberg and Arshavsky, 1997]. At the same time, the same subjects demonstrated EEG desynchronization in both hemispheres while solving arithmetic tasks. The representatives of the Western culture with the left-hemisphere cognitive set demonstrated EEG desynchronization in both hemispheres while solving any task. 7. Modern concepts of the brain laterality: a short critical reviewModern concepts of brain laterality cannot explain these data in a comprehensive way and are not free from contradictions. The first theoretical conceptualizations of brain laterality suggested that the left and right hemispheres process qualitatively different information [Goldberg and Costa, 1981 and Sperry et al., 1969]. In particular, the left hemisphere was thought to be involved in the processing of verbal material, signs, and symbols, whereas the right hemisphere was thought to be involved in handling nonverbal material, images, melodies, and spatial information. However, other researchers have refuted that point of view. In split-brain subjects, the right hemisphere is able to process verbal constructions if they are not too complicated [Ellis et al., 1988]. EEG activity in the right hemisphere predominates during the reading of stories, whereas EEG activity in the left hemisphere predominates during the reading of textbooks in science [Ornstein et al., 1979]. Although all melodies are a type of nonverbal information, the right ear (e.g., the left hemisphere) is superior to the left ear in perceiving dichotically presented melodies if they differ only in rhythm [Gordon, 1978]. Split-brain participants are partly able to report their dreams [Hoppe, 1977], even though a dream is predominantly a visual experience: even the congenitally blind individuals beyond sounds and touch sensations often have visual contents in their dreams [Bertolo et al., 2003]. The left hemisphere is superior to the right hemisphere in perceiving faces with outstanding features (such as a very long nose; [Parkin and Williamson, 1987]). Language signs for dumb people with impaired hearing are nonverbal. However, the ability to sign is damaged by the left hemisphere strokes [Bellugi et al., 1983]. According to another approach, human hemispheres differ in terms of information processing [Gordon, 1978]. That is, the left hemisphere processes verbal and nonverbal sequential information. The function of the right hemisphere is single-stage, parallel processing of many elements of information as a single whole. However, this point of view cannot account for findings that the left hemisphere is also able to grasp a series of data simultaneously and can do so as rapidly as the right hemisphere does [Polich, 1982]. According to yet another point of view [Goldberg and Costa, 1981], the left hemisphere is responsible for maintaining familiar forms of behavior, whereas the right hemisphere's specialization includes detecting novel and unexpected events. However, even that approach cannot account for all existing data. It cannot explain the advantage of the left hemisphere in identifying strange faces or the advantage of the right hemisphere in identifying normal faces with which the participant is familiar [Parkin and Williamson, 1987]. 8. Concept of mono- and polysemantic contextsIn order to avoid the abovementioned contradictions, I have presented the following concept [Rotenberg, 1979, Rotenberg, 1982, Rotenberg, 1985, Rotenberg, 1993, Rotenberg, 1994 and Rotenberg and Weinberg, 1999]. In the most general form, the difference between the two strategies of thinking related to the left- and right-hemisphere functions is reduced to opposite modes of organizing the contextual connection between elements of information. "Left-hemisphere" or formal logical thinking so organizes any sign material (whether symbolic or iconic) as to create a strictly ordered and unambiguously understood monosemantic context. Its formation requires an active choice from the many real and potential connections between the multiform objects and phenomena of a few definite connections that would not create internal contradictions and would facilitate an ordered analysis. Such a strategy of thinking makes it possible to build a pragmatically convenient but simplified model of reality. It is based on probability forecasting and a search for concrete cause-and-effect relations, and it is precisely for this model that the vector of time orientation exists. In contrast, the function of the right hemisphere is a simultaneous capture of an infinite number of connections and the formation of an integral but ambiguous polysemantic context due to this capture. In such a context, the whole is not determined by its components because all specific features of the whole are determined only by interconnections between these parts. On the contrary, any concrete element of such a context bears a determining stamp of the whole. A new experience is incorporated in this holistic picture of the world. Individual facets of images interact with each other on many semantic planes simultaneously. Examples of such contextual connections are the connections between images in sleep dreams or in works of art. The advantages of this strategy of thinking manifest themselves only when the information itself is complex, internally contradictory, and basically irreducible to an unambiguous context. The formation of the contextual connection between elements of information determines the subjects' general perception of the world and their position according to the world and belongs to the highest and most integrative brain functions. Thus, it is possible to suggest that it is the frontal lobe as the most developed structure that plays the crucial role in the formation of the context, while other brain structures are responsible for the more concrete and more restricted brain functions. Data of many experimental investigations confirm this conceptual approach either directly or indirectly. It was shown [Galaburda et al., 1978] that the right frontal lobe is larger than the left one in right-handed subjects although the left hemisphere seems to be "dominating" in cognitive functions. Moreover, arborization of the neuronal net is less prominent in the left than in the right hemisphere [Saugstad, 1998]. According to the present concept, it is possible to explain this difference: polysemantic context based on the right frontal activity integrates more facets of information and more connections between objects and events, and performs it in the most flexible way. The right hemisphere displays attention to both sides of space that is very important for the collection of all possible interrelations between facets of information, whereas the left hemisphere is sensitive only to the right side of the space [Weintraub and Mesulam, 1987]. According to the investigations of pause duration in verbal expression, [Mendonca et al., 2001] suggested an inhibitory interhemispheric function of the right hemisphere modulating verbal expression of the thought flow. Inhibition of an ongoing reaction tendency for adaptation to changing environment is also a function of the right-hemisphere prefrontal cortex [Konishi et al., 1999]. It means that the right hemisphere, being superior in reaction time [Schweinberger and Sommer, 1991] and being able to grasp any information earlier than the left hemisphere and from the broader field, may perform a function of the "censor" of consciousness and of conscious overt behavior. Such function may relate to self-image based in the right hemisphere, presumably in its frontal part [Rotenberg, 1982 and Rotenberg, 1994]. This hypothesis was recently confirmed by data that in humans the anterior right hemisphere is critically engaged in detecting the self-face and thus may selectively participate in processes linked to self-awareness [Keenan et al., 2001] as well as by data from the positron emission tomography study that the right frontal lobe is crucial for self-encoding [Craik et al., 1999]. Subjects are more accurate in solving difficult, complicated tasks when stimuli are presented in the left visual field [Tucker et al., 1999]. The right hemisphere has an advantage in processing of metaphors [Anaki et al., 1998 and Winner and Gardner, 1977]. Sense of humor is also a function of the right hemisphere [Wapner et al., 1981]. Processing of metaphors and sense of humor both require sensitivity to the polysemantic context. The hemispheric differences in meaning processing also confirm the proposed concept. The right hemisphere is able to produce a much broader net of associations than the left hemisphere. The left hemisphere activates only closely related information, while the right hemisphere activates both closely and distantly associated information to the same degree [Beeman et al., 1994]. The right hemisphere immediately activates various meanings associated with a word, while left hemisphere access is restricted to the dominant meaning [Coney and Evans, 1999]. It was shown [Chiarello, 1998, Chiarello et al., 1987 and Chiarello et al., 1990] that the left hemisphere is biased toward the processing of close lexical semantic relationships while the processing of more loosely related semantic associations relies more heavily on the right hemisphere. Inhibition of semantically unrelated elements of information in the left hemisphere corresponds to the active restriction of potential relationships between these elements proposed by my model. This model corresponds also to data [Federmeier and Kutas, 1999] suggesting that the right hemisphere is integrative and incorporates new information directly into the initial context while left hemisphere processing is predictive: new information is compared with the definite (monosemantic) expectation rather than directly with the context. The right hemisphere seems to outperform the left precisely under conditions where prediction is difficult and information is global. The left hemisphere has a more narrow––"local"––view of context. The concept of the mono- vs. polysemantic contexts corresponds to the modern view that the two hemispheres do not compete with each other but rather collaborate during the performance of difficult cognitive tasks [Banich and Belger, 1990, Reuter-Lorenz et al., 1999 and Dolcos et al., 2002]. Actually, these two types of context do not exclude one another but complete each other while dealing with different aspects of the reality. Moreover, polysemantic context has sense only in comparison to the monosemantic one: for the right hemisphere itself the world is holistic but not polysemantic. It is worth stressing that according to the results of the abovementioned investigations [De Pascalis and Palumbo, 1986, Grossman, 1988, Rotenberg and Arshavsky, 1991, Rotenberg and Arshavsky, 1997 and Whitton, 1978] it is only the organization of the left hemispheric monosemantic context that requires additional nonspecific activation of the brain from the reticular formation. The formation of the polysemantic context produced by the frontal lobe of the right hemisphere does not require any additional efforts in subjects who are initially prepared for such activity, as, for instance, in creative persons, because the polysemantic context is a natural one (numerous connections between facets of objects and phenomena is a feature of a real world). The amount of connections is not going to be artificially restricted and alpha rhythm in the right hemisphere is not decreased. In general, normal subjects demonstrate greater alpha activity in the right hemisphere than in the left [Graae et al., 1996]. However, if the formation of the polysemantic context does not depend on the physiological brain activation it is possible to assume also that the additional activation of the frontal brain cortex by the reticular formation does not help to create a polysemantic context when it is absent and cannot compensate for the functional insufficiency of the right hemispheric polysemantic way of thinking. It is necessary to emphasize once more that we are speaking only about the function of the frontal part of the right hemisphere that produces the polysemantic context. Other functions of the right hemisphere related to the occipital, temporal, and parietal structures do not display discrepancy between their efficiency and brain activation. Thus, poor spatializers (subjects who have difficulties in processing of spatial information) show higher and broader cortical activity in parietal area when efficiently processing spatial tasks [Vitouch et al., 1997]. In healthy subjects, frontal lobes are the most mature regions of the brain, and the full maturation of the right brain is the latest one [Saugstad, 1998]. It is possible to suggest that the ability to form a polysemantic context is one of the highest and the most mature brain functions. In early infancy, responses to stimuli of the environment are simple and polar––either approach or withdrawal. Thus, for infants, [Davidson, 1992] concept may be very relevant. In infants the increase of the physiological activity of the right frontal cortical/subcortical structures may correspond to withdrawal [Calkins et al., 1996]. However, it means also that in adults the increase of the physiological activation of the same structures reflects the immature behavior or regression––inability to cope with negative experience in an adaptive way by using the polysemantic way of thinking. In the context of maturation it is worth mentioning the difference between genders in brain lateralization. According to the comprehensive review of [Heller, 1993], there are only few consistent gender differences in cognitive functions, and they can be explained as an outcome of smaller differences in verbal and visuospatial performance between hemispheres in women in comparison to men (i.e., less complete lateralization). It was shown [Rescher and Rappelsberger, 1999] that for higher spatial processes the right hemisphere is more specialized in men than in women. Less complete lateralization means less specialization of hemispheres in particular functions, and less specialization is often related to impaired performance. For instance, subjects with low lateralization of speech function display an impaired performance of verbal and nonverbal tasks in comparison to persons with full lateralization [Yanson and Kenga, 1984]. However, women usually at least do not yield to men in particular functions related to the left hemisphere, and whether they are less effective in most right-hemisphere functions also seems to be very doubtful [Heller, 1993]. Thus, what functional meaning does their less complete lateralization have, and what is the reason for this relatively smoothed lateralization? Presumably, women are less lateralized in the domain of frontal brain functions, and it is related to the peculiarity of the process of brain maturation. Females in average are characterized by earlier brain maturation [Heller, 1993 and Saugstad, 1998]. It gives an advantage to the left hemisphere versus the right one because the final maturation of the left hemisphere in general appears earlier than the final maturation of the right frontal lobe, that is, as we have mentioned, the last region to mature [Saugstad, 1998]. The longer is the process of development the higher is the final functional ability of the brain structure (of course, if in the process of development the structure is not impaired). If the development of the polysemantic way of thinking is, as we suggested, related to the right frontal lobe, then the delayed maturation of this structure presents an advantage to men in this way of thinking. Thus, less complete lateralization in women may relate to the less mature and efficient polysemantic way of thinking. If this hypothesis is correct, and if polysemantic way of thinking is important for the adaptation to the polydimensional environment and for emotional equilibrium (see Section 9), then it is possible to explain the approximately 2:1 ratio of depression in women compared to men [Weissman et al., 1984]. Although women dominate in the population of depressed patients, the psychophysiological gender differences in depression are rarely taken into consideration [Heller, 1993]. Some investigations have been performed only on women, and in many other investigations females dominated. In future investigations it would be reasonable to compare the physiological activity of frontal lobes in both genders. At first glance, our hypothesis of the deficient right-hemisphere polysemantic way of thinking in women as a predisposition towards depression contradicts data that women are more lateralized than men for tasks involving a need to process emotional information and are more efficient in these tasks [Ladavas et al., 1980 and Strauss and Moscovitch, 1981]. Women's intuition in emotional relationships is a topic of legends. However, first of all, this strong intuition can be partly explained by the stronger interhemispheric transfer in females than in males [Rescher and Rappelsberger, 1999]. In addition, for women traditionally emotional relationships are more important than for men, and this importance is strengthened, even until now, by their social role and traditional social attitudes. Speaking metaphorically, for a man emotional relationships display a part of the holistic world, while for a woman the world is often a part of the emotional relationships. It can explain the high sensitivity of women just to the emotional information despite the relatively lower level of the right-hemisphere activity. This high emotional sensitivity and a predisposition towards emotional disturbances may also have a physiological background. It was shown recently [Van Elst et al., 2000] that depressed patients of both genders displayed a significant enlargement of both amygdala volumes in comparison to healthy subjects but amygdala volumes of female patients (regardless of what diagnosis they had) were increased in comparison with male patients. Of course, the possible role of the amygdala in emotional sensitivity and in the increased predisposition towards emotional disorders (including depression) does not exclude the important role of the functional right-hemisphere deficiency in the development of depression. Both brain structures are important but they seem to be independent in their contribution in depression. Depressed patients display exaggerated left, but not right, amygdala activation to fearful faces [Sheline et al., 2001]. Left amygdala metabolism was increased in unipolar depressives with familial pure depressive disease and in depressed patients with bipolar disorders and positively correlated with plasma cortisol levels [Drevets et al., 2002]. It is possible that the right-hemisphere functional insufficiency and the hyperactivity of the amygdala are two independent factors of the pathogenesis of depression. 9. The right frontal hyperactivity in depression: functional meaningIf the additional physiological activation of the frontal lobe of the right hemisphere does not help to restore its functional sufficiency and to create the polysemantic context, then it is possible to understand the co-occurence of the functional disability of the right hemisphere and its relative physiological overactivation. The latter displays the insufficient and irrelevant efforts of the brain to overcome restrictions produced by the weakness of the right-hemisphere thinking [Rotenberg and Arshavsky, 1991]. Moreover, we suggested [Rotenberg and Arshavsky, 1991 and Rotenberg and Arshavsky, 1997] that EEG desynchronization of the right hemisphere in the process of the solution of the right hemispheric tasks is a sign of the functional insufficiency of the right hemisphere. Right hemisphere activation in depression, being combined with the impaired right-hemisphere functions, confirms this suggestion. In this context it is worth stressing that activation of the right frontal region in depression and the organic damage of the same region are both accompanied by the disturbance of the right-hemisphere functions. Without the abovementioned explanation such similarity seems paradoxical. In depression EEG activity differs from that of healthy subjects in all brain regions, not only in the frontal region. Parietal and temporal brain regions of the right hemisphere are relatively less activated than the same regions of the left hemisphere [Hagemann et al., 1998 and Tucker and Dawson, 1984]. It corresponds to the impairment of the relevant functions and is an additional sign of the brain functional insufficiency in depression. By taking into consideration all the abovementioned statements, let us try to explain the role of the right-hemisphere functional insufficiency in the psychophysiological mechanisms of depression. The ability to form a polysemantic context means the ability to integrate different experiences and helps the subject to experience himself as integrated into the polydimensional and polysemantic world of objects and human interrelationships. This integration is the most important feature of the subject's mental health, emotional balance, and well-being. I suggest that as long as such integration exists, the subject is protected from mental diseases including depression, as well as from maladaptive emotions and even from psychosomatic disorders. When such integration becomes distorted due to the functional insufficiency of the right hemisphere [Rotenberg, 1995], the subject finds himself in front of a very complicated reality (including the reality of emotional relationships) full of inner contradictions. The brain then has to deal with this reality lacking an essential mechanism. At the same time, even the insufficient right hemisphere is still sensitive to the all complications and contradictions in reality, although it is unable to manage it. To accept information and to be able to deal with this information in the appropriate way are different tasks. As a result, the right hemisphere became overloaded, physiologically activated, and overwhelmed. According to this approach, the subject reacts to the negative and frustrating information by producing maladaptive depressive mood only if the adaptive mechanisms of the polysemantic way of thinking are insufficient, and the latter corresponds to the physiological activation of the frontal area of the right hemisphere. Those people who demonstrate decreased alpha activity over the right hemisphere during the baseline are less tolerant to the induction of negative affect [Tomarken et al., 1992 and Wheeler et al., 1993]. The normally functioning right hemisphere enables the subject to work through the negative affects [Schore, 1994 and Schore, 1996] and as a result prevents the development of depression. On the other hand, the functional deficiency of the right hemisphere may be a background not only of depression, but also of psychosomatic disorders [Rotenberg, 1995], schizophrenia [Rotenberg, 1994], and of suicidal behavior [Weinberg, 2000]. This functional deficiency can develop very early in life presumably due to the lack of emotional relationships with parents and primary caregivers [Rotenberg, 1982, Rotenberg, 1994 and Schore, 1996]. For instance, children who display low alpha activity over the right hemisphere cry more following separation from their mothers than children with high alpha activity over the right hemisphere [Davidson and Fox, 1989]. As we have already mentioned, a normal pleasant affect is associated with creative problem solving, flexible thinking, and an integrative organization of complicated cognitive material––all these functions based on the skills of the frontal part of the right hemisphere. In contrast, depression is characterized by the lack of cognitive flexibility and integrative strategy [Heller and Nitschke, 1997]. Thus, depression may be a consequence of the inability to integrate the complicated reality and a state of giving up in the face of this reality. The additional physiological activation of the right frontal region displays an unsuccessful effort to compensate the right-hemisphere functional deficiency and to overcome this obstacle. When subjects are unable to integrate their experience, behavior, and self-image in the most natural way––by means of the right hemispheric skills––they have only one opportunity––to try to organize it in a more formal way by means of the skills of the frontal part of the left hemisphere. Such organization is not relevant enough for the holistic reality, but it can make this reality at least subjectively more simplistic and less painful for the subject. However, depressed patients are characterized by decreased activity of the frontal part of the left hemisphere and their ability to use the highest left hemispheric skills is restricted. Even infants, who show greater than average distress during maternal separation, demonstrated lower left compared to right frontal activation [Davidson, 1992]. Such patterns of the decreased frontal left hemisphere activity represent an additional risk-factor marker of the increased vulnerability of the brain that predisposes individuals towards depression in particular conditions. This relative functional insufficiency of the left hemisphere does not allow the subject to organize information sufficiently in a monosemantic way and to replace reality with an artificially constructed world. At the same time, it protects subjects from the compensatory hyperactivation of the left hemisphere and prevents the development of hallucinations and delusions typical for schizophrenia [Rotenberg, 1994]. Fig. 1A and B presents in a schematic way the relationships between each hemisphere's functional activity and its physiological activation in normal subjects and in depressed patients.
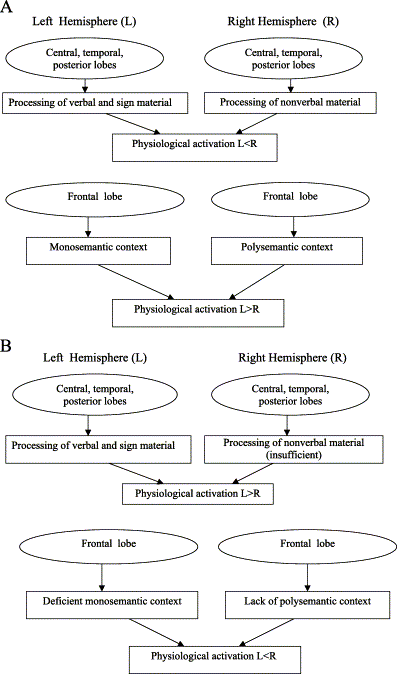
Fig. 1. (A) Hemisphere functions and psychophysiological relationships in normal subjects. (B) Hemisphere functions and psychophysiological relationships in depressed patients.
However, this does not mean that in depression the left hemisphere does not contribute to the attempts of psychological adaptation. Although restricted, this contribution is still present. This statement is confirmed by the acute exacerbation of depression after the sudden suppression of the left hemisphere. For instance, when sodium amibarbital is administered to the left carotid artery, a catastrophic reaction appears. Such initial catastrophic reaction appears even in healthy subjects presumably because the formation of the adaptive polysemantic context requires some time. Thus, just after the suppression of the left hemisphere, the subjects find themselves suddenly in front of a very complicated reality they are unable to integrate immediately––neither in a polysemantic nor, even at least partly, in a monosemantic way. Quite the contrary, the acute suppression of the right hemisphere in depressed patients usually causes a terminated decrease of depression. I suggest that in this condition the subject suddenly finds himself in front of reality that is relatively more organized by the remaining left hemispheric skills, while the suppression of the right hemisphere makes subjects less sensitive to the complications of the reality. 10. ConclusionHyperactivation of the right-hemisphere frontal part in depression reflects its functional insufficiency and inability to build a polysemantic context. In combination with the partial insufficiency of the left hemisphere frontal part, it displays a predisposition to the development of depression.

AcknowledgementsMy sincerest thanks to Prof. R. Greenberg and Dr. J. Fisch for their help and valuable comments to the first version of this manuscript.

ReferencesAdolphs et al., 2000. R. Adolphs, H. Damasio, D. Tranel, G. Coper and A.R. Damasio, A role for somatosensory cortices in the visual recognition of emotion as revealed by three-dimensional lesion mapping. J. Neurosci. 20 (2000), pp. 2683–2690. Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Alexander et al., 1996. J.E. Alexander, M.W. O'Boyle and C.P. Benbow, Developmentally advanced EEG alpha-power in gifted male and female adolescents. Int. J. Psychophysiol. 23 (1996), pp. 25–31. Abstract | Full Text + Links | PDF (644 K)
Anaki et al., 1998. D. Anaki, M. Faust and S. Kravetz, Cerebral hemispheric asymmetries in processing lexical metaphors. Neuropsychologia 36 (1998), pp. 353–362. Abstract | Abstract + References | PDF (1068 K)
Banich, 1997. M.T. Banich, Neuropsychology: The Neural Bases of Mental Function. , Houghton Mifflin, Boston (1997). Banich and Belger, 1990. M.T. Banich and A. Belger, Interhemispheric interaction: how do the hemispheres divide and conquer a task?. Cortex 26 (1990), pp. 77–94. Abstract-EMBASE
| Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Banich et al., 1992. M.T. Banich, N. Stolar, W. Heller and R. Goldman, A deficit in right hemisphere performance after induction of depressed mood. Neuropsychiatry Neuropsychol. Behav. Neurol. 5 (1992), pp. 20–27. Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
Baxter et al., 1989. L.R. Baxter, J.M. Schwartz, M.E. Phelps, J.C. Mazziotta, B.H. Guze, C.E. Selin, R.H. Gerner and R.M. Sumida, Reduction of prefrontal cortex glucose metabolism common to three types of depression. Arch. Gen. Psychiatry 46 (1989), pp. 243–250. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Beeman et al., 1994. M. Beeman, R.B. Friedman, J. Grafman, E. Perez, S. Diamond and M.B. Lindsay, Summation priming and coarse semantic coding in the right hemisphere. J. Cogn. Neurosci. 6 (1994), pp. 26–45. Abstract-PsycINFO
| Abstract-EMBASE
| $Order Document
Bellugi et al., 1983. O. Bellugi, H. Poizner and E.S. Klima, Brain organization for languages: clues from sign aphasia. Hum. Neurobiol. 2 (1983), pp. 155–170. Bench et al., 1992. C.J. Bench, K.J. Friston, R.G. Brown, L.C. Scott, R.S.J. Frackowiak and R.J. Dolan, The anatomy of melancholia: focal abnormalities of cerebral blood flow in major depression. Psychol. Med. 22 (1992), pp. 607–615. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Bench et al., 1993. C.J. Bench, K.J. Friston, R.G. Brown, R.S. Frackowiak and R.J. Dolan, Regional cerebral blood flow in depression measured by positron emission tomography: the relationship with clinical depression. Psychol. Med. 23 (1993), pp. 579–590. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Bertolo et al., 2003. H. Bertolo, T. Paiva, L. Pessoa, T. Mestre, R. Marques and R. Santos, Visual dream content, graphical representation and EEG alpha activity in congenitally blind subjects. Cognit. Brain Res. 15 (2003), pp. 277–284. Abstract | Full Text + Links | PDF (435 K)
Bowden and Beeman, 1998. E.M. Bowden and M.J. Beeman, Getting the right idea: semantic activation in the right hemisphere may help solve insight problems. Psychol. Sci. 9 (1998), pp. 435–440. Abstract-PsycINFO
| $Order Document
| Full Text via CrossRef Brody et al., 2001. A.L. Brody, S. Saxena, P. Stoessel, L.A. Gillies, L.A. Fairbanks, Alborzian, M.E. Phelps, Huang, H.-M. Wu, M.L. Ho, M.K. Ho, S.C. Au, K. Maidment and L.R. Baxter, Regional brain metabolic changes in patients with major depression treated with either paroxetine or interpersonal therapy. Arch. Gen. Psychiatry 58 (2001), pp. 631–640. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
| Full Text via CrossRef Bruder et al., 1989. G.E. Bruder, F.M. Quitkin, J.W. Stewart, C. Martin, M.M. Voglmaier and W.M. Harrison, Cerebral laterality and depression: differences in perceptual asymmetry among diagnostic subtypes. J. Abnorm. Psychology 98 (1989), pp. 177–186. Abstract | Abstract + References | PDF (1036 K)
Calkins et al., 1996. S.D. Calkins, N.A. Fox and T.R. Marshall, Behavioral and physiological antecedents of inhibited and uninhibited behavior. Child Dev. 67 (1996), pp. 523–540. Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Chiarello, 1998. C. Chiarello, On codes of meaning and the meaning of codes: semantic access and retrieval within and between hemispheres. In: M. Beeman and C. Chiarello, Editors, Right Hemisphere Language Comprehension: Perspective from Cognitive Neuroscience, Erlbaum, Mathwah, NJ (1998), pp. 141–160. Abstract-PsycINFO
| $Order Document
Chiarello et al., 1987. C. Chiarello, S. Senehi and S. Nuding, Semantic priming with abstract and concrete words: differential asymmetry may be postlexical. Brain Lang. 31 (1987), pp. 43–60. Abstract-MEDLINE
| Abstract-EMBASE
| $Order Document
Chiarello et al., 1990. C. Chiarello, C. Burgess, L. Richards and A. Pollock, Semantic and associative priming in the cerebral hemispheres: some words do, some words don't ... sometimes, some places. Brain Lang. 38 (1990), pp. 75–104. Abstract-PsycINFO
| Abstract-EMBASE
| Abstract-MEDLINE
| $Order Document
Coney and Evans, 1999. J. Coney and K.D. Evans, Hemispheric asymmetries in the resolution of lexical ambiguity. Neuropsychologia 38 (1999), pp. 272–282. Craik et al., 1999. F.I.M. Craik, T.M. Moroz, M. Moscovitch, D.T. Stuss, G. Winocur, E. Tulving and S. Kapur, In search of the self: a positron emission tomography study. Psychol. Sci. 10 (1999), pp. 26–34. Abstract-PsycINFO
| $Order Document
| Full Text via CrossRef Davidson, 1992. R.J. Davidson, Anterior cerebral asymmetry and the nature of emotion. Brain Cogn. 20 (1992), pp. 125–151. Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Davidson, 1993. R.J. Davidson, Parsing affective space: perspectives from neuropsychology and psychophysiology. Neuropsychology 7 (1993), pp. 464–475. Abstract | Abstract + References | PDF (1076 K)
Davidson, 1998. R.J. Davidson, Affective style and affective disorders. Cogn. Emot. 12 (1998), pp. 307–330. Abstract-PsycINFO
| $Order Document
| Full Text via CrossRef Davidson and Fox, 1989. R.J. Davidson and N.A. Fox, Frontal brain asymmetry predicts infants' response to maternal separation. J. Abnorm. Psychology 98 (1989), pp. 27–31. Davidson and Irwin, 1999. R.J. Davidson and W. Irwin, The functional neuroanatomy of emotion and affective style. Trends Cogn. Sci. 3 (1999), pp. 11–21. Abstract | PDF (1679 K)
Davidson et al., 1990. R.J. Davidson, P. Ekman, S.D. Saron, J.A. Senulis and W.V. Friesen, Approach–withdrawal and cerebral asymmetry: emotional expression and brain physiology I. J. Pers. Soc. Psychol. 58 (1990), pp. 330–341. Abstract | Abstract + References | PDF (2006 K)
Dawson, 1994. G. Dawson, Frontal electroencephalographic correlates of individual differences in emotion expression in infants: a brain system perspective on emotion. In: N.A. Fox, Editor, The Development of Emotion Regulation: Biological and Behavioral ConsiderationsMonographs of the Society for Research in Child Development vol. 59 (1994), pp. 135–151. Abstract-MEDLINE
| $Order Document
Debener et al., 2000. S. Debener, A. Beauducel, D. Nessler, B. Brocke, H. Heilemann and J. Kayser, Is resting anterior EEG alpha asymmetry a trait marker for depression?. Neuropsychobiology 41 (2000), pp. 31–37. Abstract-MEDLINE
| Abstract-PsycINFO
| Abstract-EMBASE
| $Order Document
| Full Text via CrossRef De Pascalis and Palumbo, 1986. V. De Pascalis and G. Palumbo, EEG alpha asymmetry: task difficulty and hypnotizability. Percept. Mot. Skills 62 (1986), pp. 139–150. Abstract-MEDLINE
| $Order Document
Dolan et al., 1993. R.J. Dolan, C.J. Bench, P.F. Liddle, K.J. Friston, C.D. Frith, P.M. Grasby and R.S. Frackowiak, Dorsolateral prefrontal cortex dysfunction in the major psychoses: symptom of disease specificity?. J. Neurol. Neurosurg. Psychiatry 56 (1993), pp. 1290–1294. Abstract-PsycINFO
| Abstract-EMBASE
| Abstract-MEDLINE
| $Order Document
Dolcos et al., 2002. F. Dolcos, H.J. Rice and R. Cabeza, Hemispheric asymmetry and aging: right hemisphere decline or asymmetry reduction. Neurosci. Biobehav. Rev. 26 (2002), pp. 819–825. Abstract | Full Text + Links | PDF (394 K)
Drevets, 1998. W.C. Drevets, Functional neuroimaging studies of depression: the anatomy of melancholia. Annu. Rev. Med. 49 (1998), pp. 341–361. Abstract-MEDLINE
| Abstract-EMBASE
| $Order Document
| Full Text via CrossRef Drevets et al., 1992. W.C. Drevets, T.O. Videen, J.L. Price, S.H. Preskorn, S.T. Carmichael and M.E. Raichle, A functional anatomical study of unipolar depression. J. Neurosci. 12 (1992), pp. 3628–3641. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
Drevets et al., 2002. W.C. Drevets, J.L. Price, M.E. Bardgett, T. Reich, R.D. Todd and M.E. Raichle, Glucose metabolism in the amygdala in depression. Relationship to diagnosis subtype and plasma cortisol levels. Pharmacol. Biochem. Behav. 71 (2002), pp. 431–447. SummaryPlus | Full Text + Links | PDF (280 K)
Ellis et al., 1985. H.C. Ellis, R.L. Thomas, A.D. McFarland and J.W. Lanie, Emotional states and retrieval in episodic memory. J. Exp. Psychol. Learn. Mem. Cogn. 11 (1985), pp. 367–370. Ellis et al., 1988. H.C. Ellis, A.W. Young and C. Andersen, Models of word recognition in the left and right hemispheres. Brain Lang. 35 (1988), pp. 254–277. Federmeier and Kutas, 1999. K.D. Federmeier and M. Kutas, Right words and left words: electrophysiological evidence for hemispheric differences in meaning processing. Cogn. Brain Res. 8 (1999), pp. 373–392. Abstract | Full Text + Links | PDF (372 K)
Field et al., 1995. T. Field, N.A. Fox, J. Pickens and T. Nawrocki, Relative right frontal EEG activation in 3- to 6-month-old infants of "depressed" mothers. Dev. Psychol. 31 (1995), pp. 358–363. Abstract | Abstract + References | PDF (502 K)
Finset, 1988. A. Finset, Depressed mood and reduced emotionality after right-hemisphere brain damage. In: M. Kinsbourne, Editor, Cerebral Hemisphere Function in Depression, American Psychiatric Press, Washington, DC (1988), pp. 49–64. Flor-Henry, 1983. P. Flor-Henry, Cerebral Basis of Psychopathology. , John Wright, Boston (1983). Gainotti, 1972. G. Gainotti, Emotional behavior and hemisphere side of lesion. Cortex 8 (1972), pp. 41–55. Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Galaburda et al., 1978. A.M. Galaburda, M. Le May and K.T.L. Kemper, Right–left asymmetries in the brain. Science 199 (1978), pp. 852–856. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Galin, 1974. D. Galin, Implications for psychiatry of left and right cerebral specialization: a neuropsychological context for unconscious process. Arch. Gen. Psychiatry 31 (1974), pp. 572–583. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
Gardner et al., 1983. H. Gardner, H.H. Brownell, W. Wapner and D. Michelow, Missing the point: the role of the right hemisphere in the processing of complex linguistic materials. In: E. Precman, Editor, Cognition Processing in the Right Hemisphere, Academic Press, New York (1983), pp. 169–191. Goldberg and Costa, 1981. E. Goldberg and L.D. Costa, Hemisphere differences in the acquisition and use of descriptive systems. Brain Lang. 14 (1981), pp. 144–173. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Gordon, 1978. H. Gordon, Left hemisphere dominants for rhythmic elements in dichotomically presented melodies. Cortex 14 (1978), pp. 58–70. Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
Graae et al., 1996. F. Graae, C. Tenke, G. Bruder, M.-J. Rotheram, J. Piacentini, D. Castro-Blanko, P. Leite and J. Towey, Abnormality of EEG alpha asymmetry in female adolescent suicide attempters. Biol. Psychiatry 40 (1996), pp. 706–713. Abstract | PDF (639 K)
Grossman, 1988. M. Grossman, Drawing deficits in brain-damaged patients freehand pictures. Brain Cogn. 8 (1988), pp. 189–205. Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Hagemann et al., 1998. D. Hagemann, E. Naumann, G. Becker, S. Maier and D. Bartussek, Frontal brain asymmetry and affective style. Psychophysiology 35 (1998), pp. 372–388. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
| Full Text via CrossRef Hagemann et al., 1999. D. Hagemann, E. Naumann, A. Lurken, G. Becker, S. Maier and D. Bartussek, EEG asymmetry, dispositional mood and personality. Pers. Individ. Differ. 27 (1999), pp. 541–568. Abstract | Full Text + Links | PDF (410 K)
Hartlage et al., 1993. S. Hartlage, L.B. Alloy, C. Vazquez and B. Dykman, Automatic and effortful processing in depression. Psychol. Bull. 113 (1993), pp. 247–278. Abstract | Abstract + References | PDF (3086 K)
Heller, 1990. W. Heller, The neuropsychology of emotion: developmental patterns and implications for psychopathology. In: N. Stein, B.L. Leventhal and T. Trabasso, Editors, Psychological and Biological Approaches to Emotion, Erlbaum, Hillsdale, NJ (1990), pp. 167–211. Abstract-PsycINFO
| $Order Document
Heller, 1993. W. Heller, Gender differences in depression: perspectives from neuropsychology. J. Affect. Disord. 29 (1993), pp. 129–143. Abstract | Abstract + References | PDF (1577 K)
Heller and Nitschke, 1997. W. Heller and J.B. Nitschke, Regional brain activity in emotions: a framework for understanding cognition in depression. Cogn. Emot. 11 (1997), pp. 637–661. Abstract-PsycINFO
| $Order Document
Heller and Nitschke, 1998. W. Heller and J.B. Nitschke, The puzzle of regional brain activity in depression and anxiety: the importance of subtypes and comorbidity. Cogn. Emot. 12 (1998), pp. 421–447. Abstract-PsycINFO
| $Order Document
| Full Text via CrossRef Henriques and Davidson, 1990. J. Henriques and R. Davidson, Regional brain electrical asymmetries discriminate between previously depressed and healthy control subjects. J. Abnorm. Psychol. 99 (1990), pp. 23–31. Hirai, 1974. T. Hirai, Psychophysiology of Zen. , Igako Shoin, Tokyo (1974). Hoppe, 1977. K.D. Hoppe, Split brain and psychoanalysis. Psychoanal. Q. 46 (1977), pp. 220–242. Isen et al., 1982. A.M. Isen, B. Means, R. Patrick and G. Nowicki, Some factors influencing decision-making strategy and risk taking. In: M.S. Clark and S.J. Fiska, Editors, Affect and Cognition, Erlbaum, Hillsdale, NJ (1982), pp. 213–261. Jacobs and Snyder, 1996. G.D. Jacobs and D. Snyder, Frontal brain asymmetry predicts affective style in men. Behav. Neurosci. 110 (1996), pp. 3–6. Abstract | Abstract + References | PDF (292 K)
Jones-Gotman and Milner, 1977. M. Jones-Gotman and B. Milner, Design fluency: the invention of nonsense drawings after focal cortical lesions. Neuropsychologia 15 (1977), pp. 653–674. Abstract | Abstract + References | PDF (1481 K)
Karpinis et al., 1995. G. Karpinis, J. Nimatoudis, A. Karavatos, D. Kandylis and S. Karpinis, Functional brain organization in bipolar affective patients during manic phase and after recovery: a digit dichotic listening study. Percept. Mot. Skills 80 (1995), pp. 1275–1282. Kasumoto and Hirai, 1966. A. Kasumoto and T. Hirai, An electroencephalographic study of Zen meditation. Folia Psychiatr. Neurol. Jpn. 20 (1966), pp. 315–336. Keenan et al., 2001. J.P. Keenan, A. Nelson, M. O'Connor and A. Pascual-Leone, Self-recognition and the right hemisphere. Nature 409 (2001), p. 305. Abstract-Elsevier BIOBASE
| Abstract-EMBASE
| Abstract-MEDLINE
| $Order Document
| Full Text via CrossRef Konishi et al., 1999. S. Konishi, K. Nakajima, I. Uchida, H. Kikyo, M. Kameyama and Y. Miyashita, Common inhibitory mechanism in human inferior prefrontal cortex revealed by event-related functional MRI. Brain 122 (1999), pp. 981–991. Abstract-EMBASE
| Abstract-MEDLINE
| Abstract-Elsevier BIOBASE
| $Order Document
| Full Text via CrossRef Kronfol et al., 1978. Z. Kronfol, K.S. de Hamsher and K. Digire, Depression and hemisphere functions: changes associated with unilateral ECT. Br. J. Psychiatry 132 (1978), pp. 560–567. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Kuper, 1988. J. Kuper, Editor, A Lexicon of Psychology, Psychiatry and Psychoanalysis, Routledge, London (1988), pp. 102–106. Ladavas et al., 1980. E. Ladavas, C. Umilta and P.E. Ricci-Bitti, Right hemisphere interference during negative affect: a reaction time study. Neuropsychologia 22 (1980), pp. 479–485. Ladavas et al., 1984. E. Ladavas, R. Nicoletti, C. Umilta and G. Rizzolatti, Right hemisphere interference during negative affect: a reaction time study. Neuropsychologia 22 (1984), pp. 479–485. Abstract | Abstract + References | PDF (599 K)
Lee et al., 1988. G.P. Lee, D.W. Loring, K.J. Meador, H.F. Flanigin and B.S. Brooks, Severe behavioral complications following intracarotid sodium amobarbital injection: implications for hemispheric asymmetry of emotion. Neurology 38 (1988), pp. 1233–1236. Abstract-EMBASE
| Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Levy, 1983. J. Levy, Individual differences in cerebral hemisphere asymmetry. Theoretical issues and experimental considerations. In: J.B. Hellige, Editor, Cerebral Hemisphere Asymmetry: Method, Theory and Application, Praeger, New York (1983), pp. 465–515. Lindsley, 1951. D.B. Lindsley, Emotion. In: S.S. Stevens, Editor, Handbook of Experimental Psychology, Academic Press, New York (1951), pp. 473–516. Abstract-PsycINFO
| $Order Document
Liotti and Mayberg, 2001. M. Liotti and H.S. Mayberg, The role of functional neuroimaging in the neuropsychology of depression. J. Clin. Exp. Neuropsychol. 23 (2001), pp. 121–136. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
| Full Text via CrossRef Liotti et al., 1988. M. Liotti, D. Sava and P. Cafara, Hemispheric differences in patients affected by depression and anxiety. In: M. Kinsbourne, Editor, Cerebral Hemisphere Function in Depression, American Psychiatric Press, Washington, DC (1988), pp. 99–131. Livanov and Sviderskaya, 1984. M.N. Livanov and N.E. Sviderskaya, Psychological aspects of the phenomenon of the spatial synchronization of biopotentials. Psychol. J. 5 (1984), pp. 71–83. Abstract-PsycINFO
| $Order Document
Mandal et al., 1999. M.K. Mandal, J.C. Borod, H.S. Asthana, A. Mohanty, S. Mohanty and E. Koff, Effects of lesion variables and emotion type on the perception of facial emotion. J. Nerv. Ment. Dis. 187 (1999), pp. 603–609. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
| Full Text via CrossRef Martin et al., 2001. S.D. Martin, E. Martin, S.S. Rai, M.A. Richardson and R. Royal, Brain blood flow changes in depressed patients treated with interpersonal psychotherapy or venlafaxine hydrochloride. Arch. Gen. Psychiatry 58 (2001), pp. 641–648. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
| Full Text via CrossRef Martindale, 1975. C. Martindale, What makes creative people different?. Psychol. Today, July (1975), pp. 44–48. Mayberg et al., 1999. H.S. Mayberg, M. Liotti, S.K. Brannan, S. McGinnes, R.K. Mahurin, P.A. Jerabek, J.A. Silva, J.L. Tekkel, Martin, J.L. Lancaster and P.T. Fox, Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am. J. Psychiatr. 156 (1999), pp. 675–682. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Mendonca et al., 2001. P.B. Mendonca, L.C. Piccinin, C.M. Capucho and C.J. Campos, Effect of lateralized epileptic discharges on the thought flow. Arq. Neuro-Psiquiatr. 59 (2001), pp. 318–323. Miller et al., 1995. E.N. Miller, T.A.T. Fujioka, L.J. Chapman and J.P. Chapman, Hemispheric asymmetries of function in patient with major affective disorders. J. Psychiatr. Res. 29 (1995), pp. 173–183. Abstract | Abstract + References | PDF (723 K)
Ornstein, 1972. R. Ornstein, The Psychology of Consciousness. , Freeman, San Francisco (1972). Ornstein et al., 1979. R. Ornstein, J. Herron, J. Johnstone and C. Swencionis, Differential right hemisphere involvement in two reading tasks. Psychophysiology 16 (1979), pp. 398–401. Abstract-PsycINFO
| Abstract-EMBASE
| Abstract-MEDLINE
| $Order Document
Parkin and Williamson, 1987. A.J. Parkin and P. Williamson, Cerebral lateralization at different stages of facial processing. Cortex 23 (1987), pp. 99–110. Abstract-PsycINFO
| Abstract-EMBASE
| Abstract-MEDLINE
| $Order Document
Perria et al., 1961. L. Perria, G. Rossadini and G.F. Rossi, Determination of side of cerebral dominance with amobarbital. Arch. Neurol. 4 (1961), pp. 173–181. Perris et al., 1981. C. Perris, L. von Knorring, J. Cumberbach and F. Marciano, Further studies of depressed patients by means of computerized EEG. Adv. Biol. Psychiatry 6 (1981), pp. 41–49. Polich, 1982. J.M. Polich, Hemispheric differences for visual search: serial vs. parallel processing revisited. Neuropsychologia 20 (1982), pp. 297–307. Abstract | Abstract + References | PDF (952 K)
Rescher and Rappelsberger, 1999. B. Rescher and P. Rappelsberger, Gender dependent EEG-changes during a mental rotation task. Int. J. Psychophysiol. 33 (1999), pp. 209–222. Abstract | Full Text + Links | PDF (224 K)
Reuter-Lorenz et al., 1999. P.A. Reuter-Lorenz, L. Stanczak and A.C. Miller, Neural recruitment and cognitive aging: two hemispheres are better than one, especially as you age. Psychol. Sci. 10 (1999), pp. 494–500. Rotenberg, 1979. V.S. Rotenberg, Word and image: the problem of context. Dyn. Psychiatr. 59 (1979), pp. 494–498. Abstract-EMBASE
| $Order Document
Rotenberg, 1982. V.S. Rotenberg, Funktionale Dichotomie der Gehirnhemispheren und die Bedeutung der Suchaktivitat fur Physiologische und psychopathologische Processe. In: G. Ammon, Editor, Hanbuch Der Dynamische Psychiatrie vol. 2, Ernst Reinhardt Verlag, Munchen (1982), pp. 275–335. Rotenberg, 1985. V.S. Rotenberg, Sleep dreams, cerebral dominance and creation. Pavlovian J. Biol. Sci. 20 (1985), pp. 53–58. Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Rotenberg, 1993. V.S. Rotenberg, Richness against freedom: two hemisphere functions and the problem of creativity. Eur. J. High Ability 4 (1993), pp. 11–19. Rotenberg, 1994. V.S. Rotenberg, An integrative psychophysiological approach to brain hemisphere functions in schizophrenia. Neurosci. Biobehav. Rev. 18 (1994), pp. 487–495. Abstract | Abstract + References | PDF (1193 K)
Rotenberg, 1995. V.S. Rotenberg, Right hemisphere insufficiency and illness in the context of search activity concept. Dyn. Psychiatr. 150/151 (1995), pp. 54–63. Rotenberg and Arshavsky, 1991. V.S. Rotenberg and V.V. Arshavsky, Psychophysiology of hemispheric asymmetry. The "entropy" of right hemisphere activity. Integr. Physiol. Behav. Sci. 26 (1991), pp. 183–188. Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Rotenberg and Arshavsky, 1997. V.S. Rotenberg and V.V. Arshavsky, Right and left brain hemisphere activation in the representatives of two different cultures. Homeostasis 38 (1997), pp. 49–57. Abstract-PsycINFO
| $Order Document
Rotenberg and Weinberg, 1999. V.S. Rotenberg and I. Weinberg, Human memory, cerebral hemisphere and the limbic system: a new approach. Genet. Soc. Gen. Psychol. Monogr. 125 (1999), pp. 45–70. Abstract-MEDLINE
| Abstract-PsycINFO
| $Order Document
Saugstad, 1998. L.F. Saugstad, Cerebral lateralization and rate of maturation. Int. J. Psychophysiol. 28 (1998), pp. 37–62. Abstract | PDF (2137 K)
Schaffer et al., 1983. C.E. Schaffer, R.J. Davidson and C. Saron, Frontal and parietal electroencephalogram asymmetry in depressed and non-depressed subjects. Biol. Psychiatry 18 (1983), pp. 753–762. Abstract-MEDLINE
| Abstract-PsycINFO
| Abstract-EMBASE
| $Order Document
Schore, 1994. A.N. Schore, Affect Regulation and the Origin of the Self. The Neurobiology of Emotional Development. , Erlbaum, Hillsdale, NJ (1994). Schore, 1996. A.N. Schore, The experience-dependent maturation of a regulatory system in the orbital prefrontal cortex and the original of developmental psychopathology. Dev. Psychopathol. 8 (1996), pp. 59–87. Abstract-PsycINFO
| $Order Document
Schwartz et al., 1975. G.E. Schwartz, R.J. Davidson and F. Maer, Right hemisphere lateralization for emotion in the human brain: interactions with cognition. Science 19 (1975), pp. 286–288. Abstract-MEDLINE
| Abstract-PsycINFO
| Abstract-EMBASE
| $Order Document
Schweinberger and Sommer, 1991. S.R. Schweinberger and W. Sommer, Contributions in stimulus encoding and memory search to right hemisphere superiority in face recognition: behavioural and electrophysiological evidence. Neuropsychologia 29 (1991), pp. 389–413. Abstract | Abstract + References | PDF (1966 K)
Sheline et al., 2001. Y.I. Sheline, D.M. Barch, J.M. Donnelly, J.M. Ollinger, A.Z. Snyder and M.A. Mintun. Biol. Psychiatry 50 (2001), pp. 651–658. Abstract | Full Text + Links | PDF (290 K)
Soares and Mann, 1997. J.C. Soares and J.J. Mann, The functional neuroanatomy of mood disorders. J. Psychiatr. Res. 31 (1997), pp. 393–432. Abstract | Abstract + References | PDF (2727 K)
Sobotka et al., 1992. S.S. Sobotka, R.J. Davidson and J.A. Senulis, Anterior brain electrical asymmetries in response to reward and punishment. Electroencephal. Clin. Neurophysiol. 83 (1992), pp. 236–247. Abstract | Abstract + References | PDF (1115 K)
Sperry et al., 1969. R. Sperry, M. Gazzaniga and I. Bogen, Interhemispheric relationships: the neocortical commissures, syndromes of hemisphere disconnection. In: P.O. Vinken and G.W. Bruyn, Editors, Handbook of Clinical Neurology, North Holland, Amsterdam (1969), pp. 273–290. Starkstein and Robinson, 1988. S.E. Starkstein and R.G. Robinson, Lateralized emotional response following stroke. In: M. Kinsbourne, Editor, Cerebral Hemisphere Function in Depression, American Psychiatric Press, Washington, DC (1988), pp. 25–47. Stigsby et al., 1981. B. Stigsby, J.C. Rodenberg and H.B. Moth, Electroencephalographic findings during mantra meditation. Electroencephalogr. Clin. Neurophysiol. 51 (1981), pp. 434–442. Abstract | Abstract + References | PDF (590 K)
Strauss and Moscovitch, 1981. E. Strauss and M. Moscovitch, Perception of facial expressions. Brain Lang. 13 (1981), pp. 308–332. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Swenson and Tucker, 1983. R.A. Swenson and D.M. Tucker, Lateralized cognitive style and self-description. Int. J. Neurosci. 21 (1983), pp. 91–100. Tomarken et al., 1990. A.J. Tomarken, R.J. Davidson and J.B. Henriques, Resting frontal brain asymmetry predicts affective responses to films. J. Pers. Soc. Psychol. 59 (1990), pp. 791–801. Abstract | Abstract + References | PDF (1237 K)
Tomarken et al., 1992. A.J. Tomarken, R.J. Davidson, R.E. Wheeler and R.C. Doss, Individual differences in anterior brain asymmetry and fundamental dimensions of emotion. J. Pers. Soc. Psychol. 62 (1992), pp. 676–682. Tucker, 1981. D.M. Tucker, Lateral brain function, emotion and conceptualization. Psychol. Bull. 89 (1981), pp. 19–46. Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Tucker, 1988. D.M. Tucker, Neuropsychological mechanisms of affective self-regulating. In: M. Kinsbourne, Editor, Cerebral Hemisphere Function in Depression, American Psychiatric Press, Washington, DC (1988), pp. 99–131. Tucker and Dawson, 1984. D.M. Tucker and S.L. Dawson, Asymmetric EEG power and coherence as method actors generated emotions. Biol. Psychol. 19 (1984), pp. 63–75. Abstract | Abstract + References | PDF (945 K)
Tucker et al., 1981. D.M. Tucker, C.E. Stensiel, R.S. Roth and S.L. Shearer, Right frontal lobe activation and right hemisphere performance. Arch. Gen. Psychiatry 38 (1981), pp. 169–174. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
Tucker et al., 1999. D.M. Tucker, A. Hartry-Speiser, L. McDougal, Luu and D. de Grandpre, Mood and spatial memory: emotion and right hemisphere contribution to spatial cognition. Biol. Psychiatry 50 (1999), pp. 103–125. Abstract | Full Text + Links | PDF (326 K)
Van Elst et al., 2000. L.T. Van Elst, F. Woermann, L. Lemieux and M.R. Trimble, Increased amygdala volumes in female and depressed humans. A quantitative magnetic resonance imaging study. Neurosci. Lett. 281 (2000), pp. 103–106. Vitouch et al., 1997. O. Vitouch, H. Bauer, G. Gittler, M. Leodolter and U. Leodolter, Cortical activity of good and poor spatial test performers during spatial and verbal processing studied with slow potential topography. Int. J. Psychophysiol. 27 (1997), pp. 183–199. Abstract | Full Text + Links | PDF (1777 K)
Wapner et al., 1981. W. Wapner, S. Hamby and H. Gardner, The role of the right hemisphere in the apprehension of complex linguistic materials. Brain Lang. 14 (1981), pp. 15–33. Abstract-EMBASE
| Abstract-PsycINFO
| Abstract-MEDLINE
| $Order Document
Weinberg, 2000. I. Weinberg, The prisoners of despair: right hemisphere deficiency and suicide. Neurosci. Biobehav. Rev. 24 (2000), pp. 799–815. Abstract | Full Text + Links | PDF (139 K)
Weingartner et al., 1981. H. Weingartner, R. Cohen, D. Murphy, J. Martello and C. Gerdt, Cognitive processes in depression. Arch. Gen. Psychiatry 38 (1981), pp. 42–47. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
Weintraub and Mesulam, 1987. S. Weintraub and M.M. Mesulam, Right hemisphere dominance in spatial attention. Arch. Neurol. 44 (1987), pp. 621–625. Abstract-PsycINFO
| Abstract-MEDLINE
| Abstract-EMBASE
| $Order Document
Weissman et al., 1984. M.M. Weissman, P.J. Leaf, C.E. Holzer, III, J.K. Myers and G.L. Tischler, The epidemiology of depression: an update on sex differences in rates. J. Affect. Disord. 7 (1984), pp. 179–188. Abstract | Abstract + References | PDF (420 K)
Wertheim, 1974. A.H. Wertheim, Oculomotor control and occipital alpha activity. A review and a hypothesis. Acta Psychol. 88 (1974), pp. 235–256. Abstract | Abstract + References | PDF (1065 K)
Wheeler et al., 1993. R.E. Wheeler, R.J. Davidson and A.J. Tomarken, Frontal brain asymmetry and emotional reactivity: a biological substrate of affective style. Psychophysiology 30 (1993), pp. 82–89. Abstract-MEDLINE
| Abstract-EMBASE
| Abstract-PsycINFO
| $Order Document
Whitton, 1978. T. Whitton, EEG frequency patterns associated with hallucinations in schizophrenics and creativity in normals. Biol. Psychiatry 13 (1978), pp. 123–133. Winner and Gardner, 1977. E. Winner and H. Gardner, The comprehension of metaphor in brain damaged patients. Brain 100 (1977), pp. 717–729. Abstract-MEDLINE
| Abstract-EMBASE
| $Order Document
Wu et al., 1999. J. Wu, M.S. Buchsbaum, Gillin, Tang, S. Gadwell, M. Wiegand, A. Najafi, E. Klein, K. Hazen and W.E. Bunney, Prediction of antidepressant effects of sleep deprivation by metabolic rates in the ventral anterior cingulate and medial prefrontal cortex. Am. J. Psychiatr. 156 (1999), pp. 1149–1158. Abstract-MEDLINE
| Abstract-PsycINFO
| Abstract-EMBASE
| $Order Document
Yanson and Kenga, 1984. V.N. Yanson and Z.G. Kenga, The dynamic of EEG alpha rhythm and lateralization of speech. Izv. Akad. Nauk Latv. SSR 10 (1984), pp. 103–107.
|